CORRECTING and REPLACING Vivimed Life Sciences Pvt Ltd Issues Voluntary Nationwide Recall of Losartan Potassium 25 mg, 50 mg and 100 mg Tablets, USP Due to the Detection of Trace Amounts of

These highlights do not include all the information needed to use LOSARTAN POTASSIUM TABLETS USP safely and effectively. See full prescribing information for LOSARTAN POTASSIUM TABLETS USP.LOSARTAN POTASSIUM tablets USP, for oral

CORRECTING and REPLACING Vivimed Life Sciences Pvt Ltd Issues Voluntary Nationwide Recall of Losartan Potassium 25 mg, 50 mg and 100 mg Tablets, USP Due to the Detection of Trace Amounts of

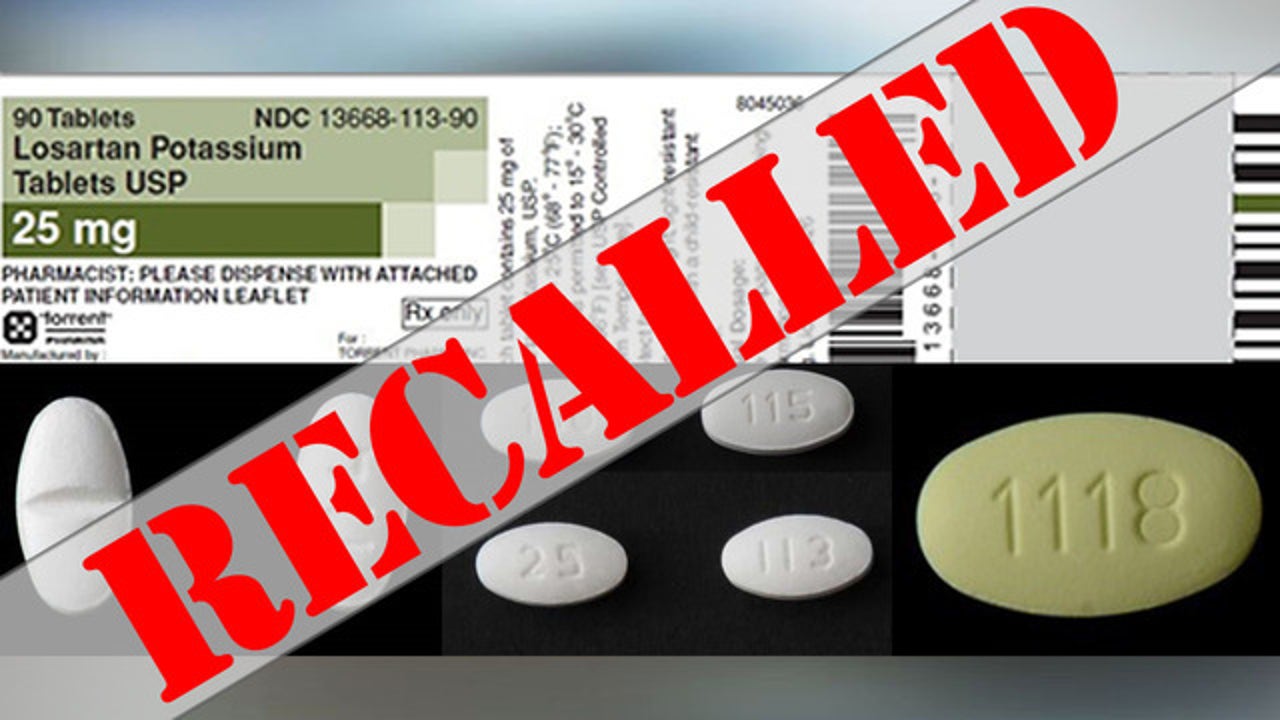
UPDATED: Torrent Pharmaceuticals Limited Issues Voluntary Nationwide Recall of Losartan Potassium Tablets, USP and Losartan Potassium and Hydrochlorothiazide Tablets, USP | FDA




/cloudfront-us-east-1.images.arcpublishing.com/gray/FATG3HNHYND73AYZFXFQNG5BBM.png)