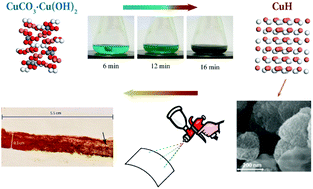
Synthesis of copper hydride (CuH) from CuCO3·Cu(OH)2 – a path to electrically conductive thin films of Cu - Dalton Transactions (RSC Publishing)
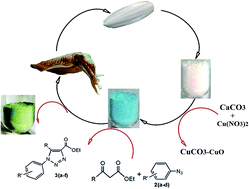
CuCO3–CuO nanocomposite as a novel and environmentally friendly catalyst for triazole synthesis - RSC Advances (RSC Publishing)
![Oxidised copper pipe. Is this Copper Carbonate [CuCO3] or is this Basic Copper Carbonate [Cu2(OH)2 CO3] : r/chemistry Oxidised copper pipe. Is this Copper Carbonate [CuCO3] or is this Basic Copper Carbonate [Cu2(OH)2 CO3] : r/chemistry](https://i.redd.it/hyb6udu06m471.jpg)
Oxidised copper pipe. Is this Copper Carbonate [CuCO3] or is this Basic Copper Carbonate [Cu2(OH)2 CO3] : r/chemistry

Copper Carbonate,Senior Chemistry - Extended Experimental In-Industry News-Nickel Acetate,Cobalt Sulfate-Fairsky Industrial Co., Limited
![SOLVED: CuCO3 is a sparingly soluble salt with a Ksp of 2.30x10^-10. The addition of NH3(aq) to CuCO3(s) yields the complex ion [Cu(NH3)4]2+(aq) with a Kf of 1.07x10^12. a. Write out the SOLVED: CuCO3 is a sparingly soluble salt with a Ksp of 2.30x10^-10. The addition of NH3(aq) to CuCO3(s) yields the complex ion [Cu(NH3)4]2+(aq) with a Kf of 1.07x10^12. a. Write out the](https://cdn.numerade.com/ask_previews/777635c8-9180-425e-8282-859f6e9ef7ec_large.jpg)
SOLVED: CuCO3 is a sparingly soluble salt with a Ksp of 2.30x10^-10. The addition of NH3(aq) to CuCO3(s) yields the complex ion [Cu(NH3)4]2+(aq) with a Kf of 1.07x10^12. a. Write out the

Green color CuCO3 for Wood Preservation Copper (II) Carbonate Basic - China Copper, Copper Carbonate | Made-in-China.com





![BASIC COPPER CARBONATE, 1 lb , [CuCO3]&[Cu(OH)2], 98% - 100% | eBay BASIC COPPER CARBONATE, 1 lb , [CuCO3]&[Cu(OH)2], 98% - 100% | eBay](https://i.ebayimg.com/images/g/KGAAAOSwkW5eLeia/s-l1200.jpg)