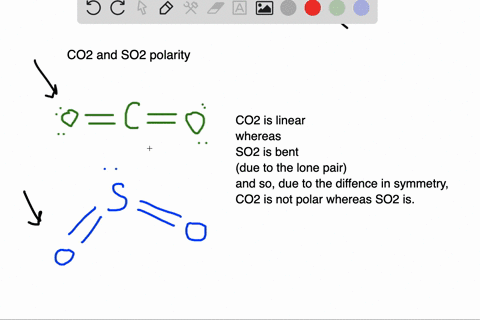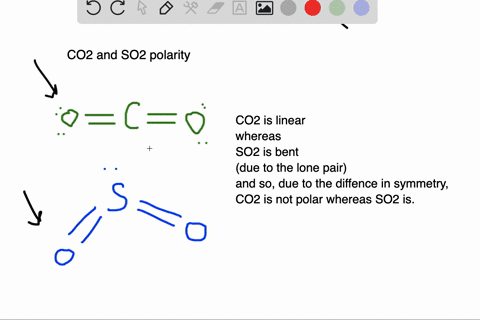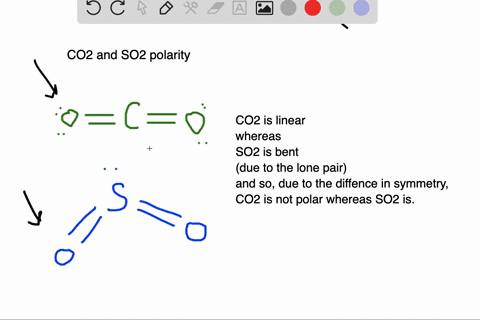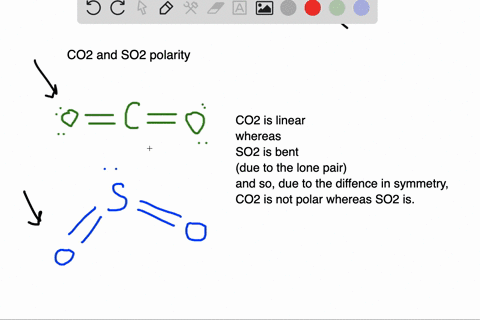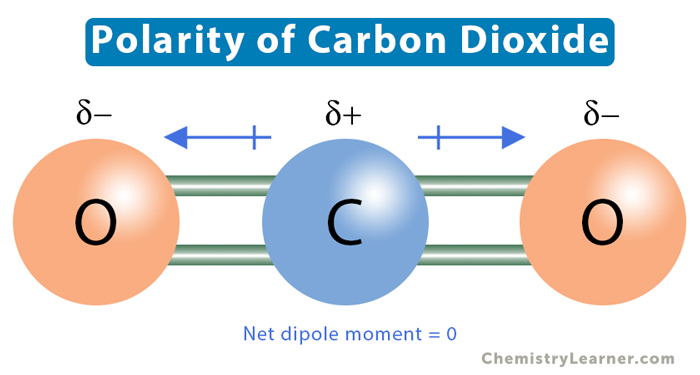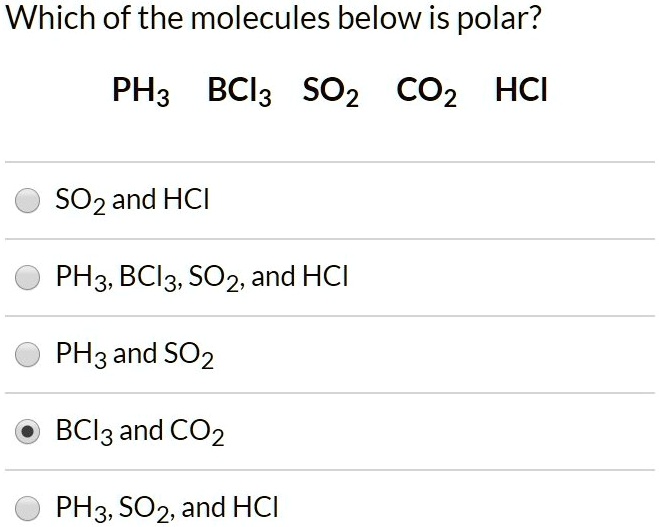
SOLVED: Which of the molecules below is polar? PH3 BCl3 SO2 CO2 HCI SO2 and HCI PH3, BCl3, SO2, and HCI PH3 and SO2 BCl3 : and CO2 PH3 SO2, and HCI

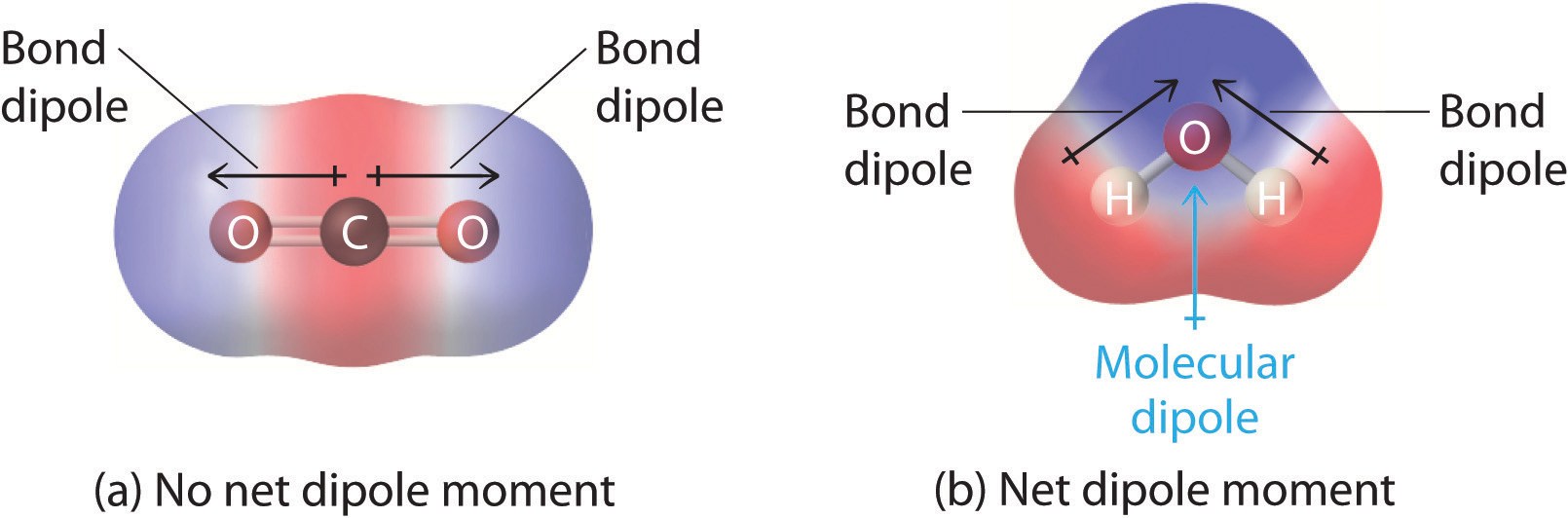
Must polar bonds give rise to polar molecules? And, why is water a polar molecule? | anhourofchemaday
Similar H2O consists of polar molecules. CO2 consists of nonpolar molecules. How do chemists explain this difference? - Quora